Medtronic Secures CE Mark for Stealth AXiS™ Surgical System, Advancing AI-Powered Spine and Cranial Care in Europe
Next-generation platform integrates planning, navigation, and robotics into a single intelligent system, enabling real-time surgical insights and scalable adoption across specialties
Image Courtesy: Public Domain
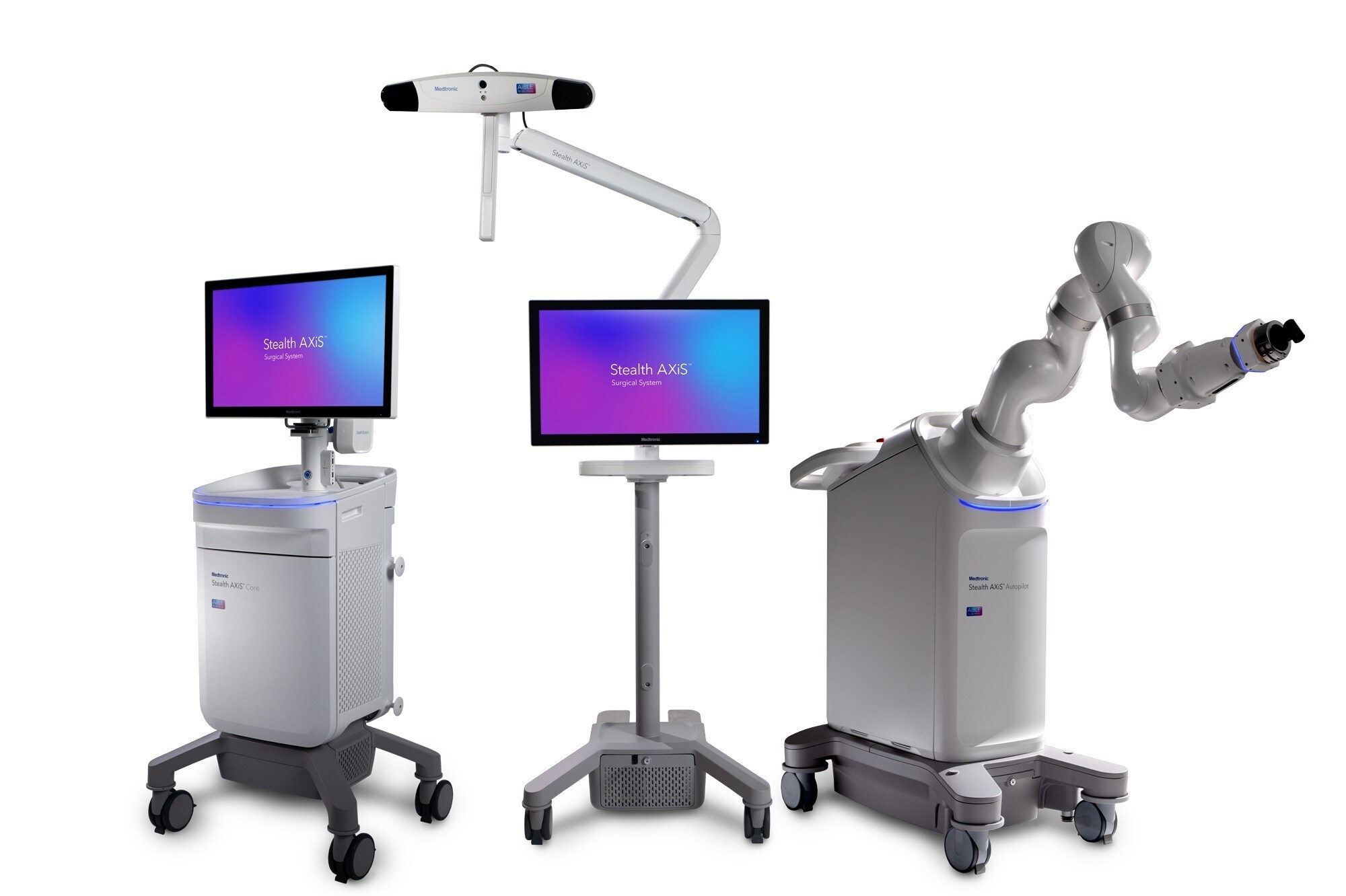
Medtronic, a global leader in healthcare technology, has received CE mark for the Stealth AXiS™ surgical system, a next-generation platform that brings planning, navigation, and robotics together into one single, intelligent system. The Stealth AXiS™ system is indicated for spine and cranial procedures, highlighting the platform's scalability across multiple surgical specialties for Medtronic.
"Receiving CE mark for the Stealth AXiS™ system marks an important milestone in expanding access to advanced surgical technologies across Europe," said Oli Prosperi, Vice President, International Commercial, Cranial & Spinal Technologies, Medtronic. "Achieving CE mark in the current regulatory environment, under EU Medical Device Regulation (MDR), underscores our commitment to bringing meaningful innovation into a highly diverse healthcare landscape."
The Stealth AXiS™ system represents major advancement in surgical technology by integrating planning, navigation, and robotic execution into a single, modular platform. Designed to support a wide range of clinical workflows and surgical environments, the system enables hospitals to adopt capabilities based on their needs, while maintaining flexibility for future expansion.
A key innovation of the system is LiveAlign™ segmental tracking, an industry-first capability that allows surgeons to visualize anatomical motion and patient alignment in real time during spine procedures. As a cornerstone of Medtronic's AiBLE™ ecosystem, the Stealth AXiS™ system connects pre-, intra-, and post-operative workflows, enabling a more integrated approach to surgical care.
A key strength of the platform lies in its AI-driven architecture, which enables advanced planning and visualization across all stages of surgery - before, during, and after the procedure. In cranial applications, it features AI-powered automatic tractography, allowing surgeons to create patient-specific brain maps and clearly visualize critical neural pathways linked to essential brain functions.
"The introduction of the Stealth AXiS™ system to Europe reflects our focus on delivering connected platforms that improve precision, efficiency, and consistency across diverse healthcare systems," said Michael Carter, Senior Vice President and President of Cranial & Spinal Technologies at Medtronic. "By bringing planning, navigation, and robotic guidance into a unified workflow, we are helping surgeons translate insight into action in the operating room, supporting more predictable and personalized care while building a strong foundation for continued innovation."
The CE mark follows U.S. FDA clearances earlier this year, highlighting Medtronic's ability to rapidly bring advanced surgical technologies to global markets.