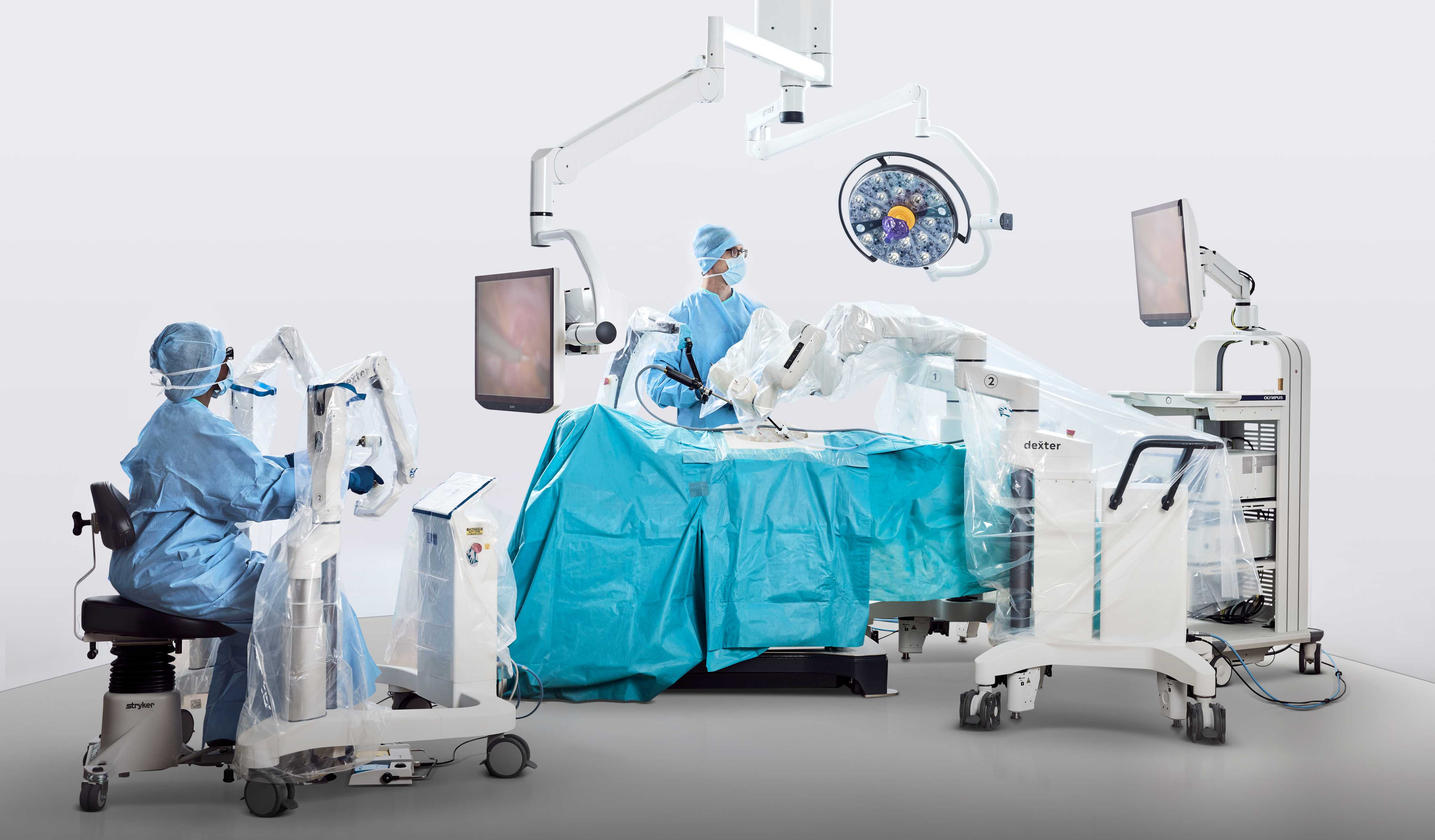
Distalmotion Submits FDA 510(k) for Expanded Gynecologic Indications for DEXTER® Robotic Surgery System
Filing with the U.S. Food and Drug Administration aims to extend DEXTER’s use in sacrocolpopexy, sacrocervicopexy, and endometriosis procedures, strengthening robotic surgery adoption in outpatient settings.
Image Courtesy: Public Domain
Distalmotion, the global MedTech company expanding access to robotic surgery in outpatient sites of care, announced that it submitted a 510(k) application to the US Food and Drug Administration (FDA) in March to seek clearance for sacrocolpopexy, sacrocervicopexy, and endometriosis resection for the DEXTER® Robotic Surgery System in the US.
This latest submission follows the completion of the Robotic Sacrocolpopexy and Sacrocervicopexy (SPARO) clinical trial evaluating DEXTER in these procedures. If cleared, this would represent the fourth overall indication and second in gynecology for DEXTER in the US and further support the expansion of service lines in ambulatory surgery centers (ASCs).
As procedures continue to shift from hospitals to outpatient settings, driven by physician, payor, and patient preferences, robotics has become a key enabler of this migration. For ASCs building gynecology programs, expanded robotic indications allow multiple procedure types—such as hysterectomy, sacrocolpopexy, and other benign gynecologic procedures—to be performed within the same operating day, supporting more efficient service line operations.
“Expanding gynecology capabilities in outpatient environments represents an important next step in the evolution of the DEXTER robotic program,” said Greg Roche, CEO of Distalmotion. “DEXTER is designed to help outpatient centers introduce robotics in a practical way, enabling care teams to build sustainable service lines across multiple specialties.”
Distalmotion also received FDA approval for a US myomectomy trial via Investigational Device Exemption (IDE) to further advance its clinical work in gynecology. Myomectomy represents a large procedure segment in US gynecologic surgery, with this market estimated to reach approximately $1 billion by 2030[1].
“DEXTER is purpose-built for ASCs, and our expansion of general surgery and gynecology indications supports the delivery of the combined benefits of wristed robotics and same-day care in a cost-effective way for patients, physicians, and providers alike. This submission reflects our commitment to enabling access to robotic surgery in the ASC environment,” said Mr. Roche.